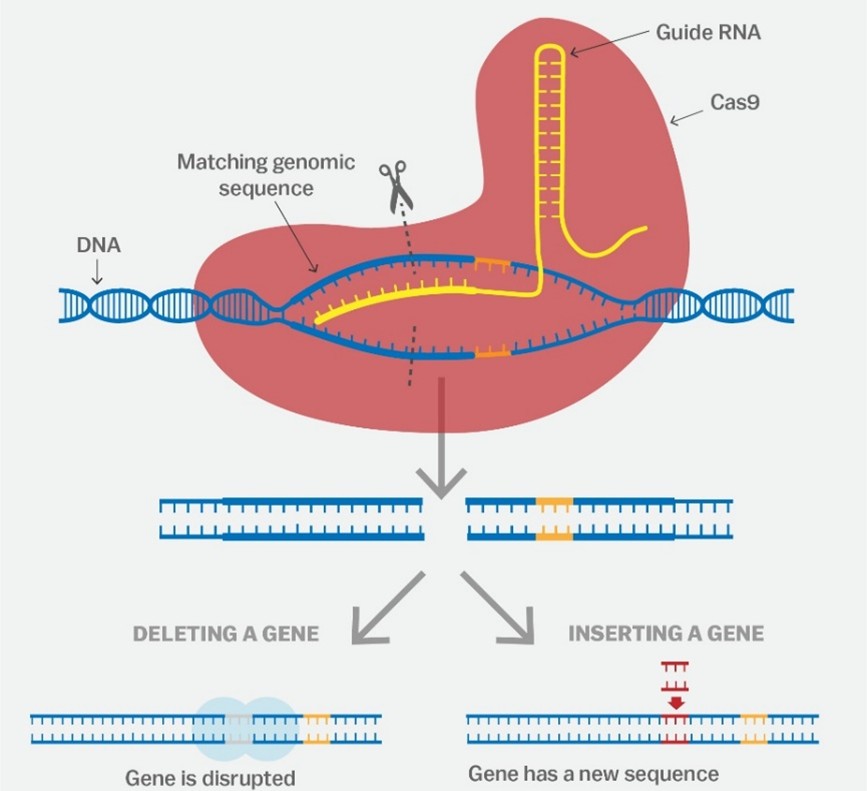
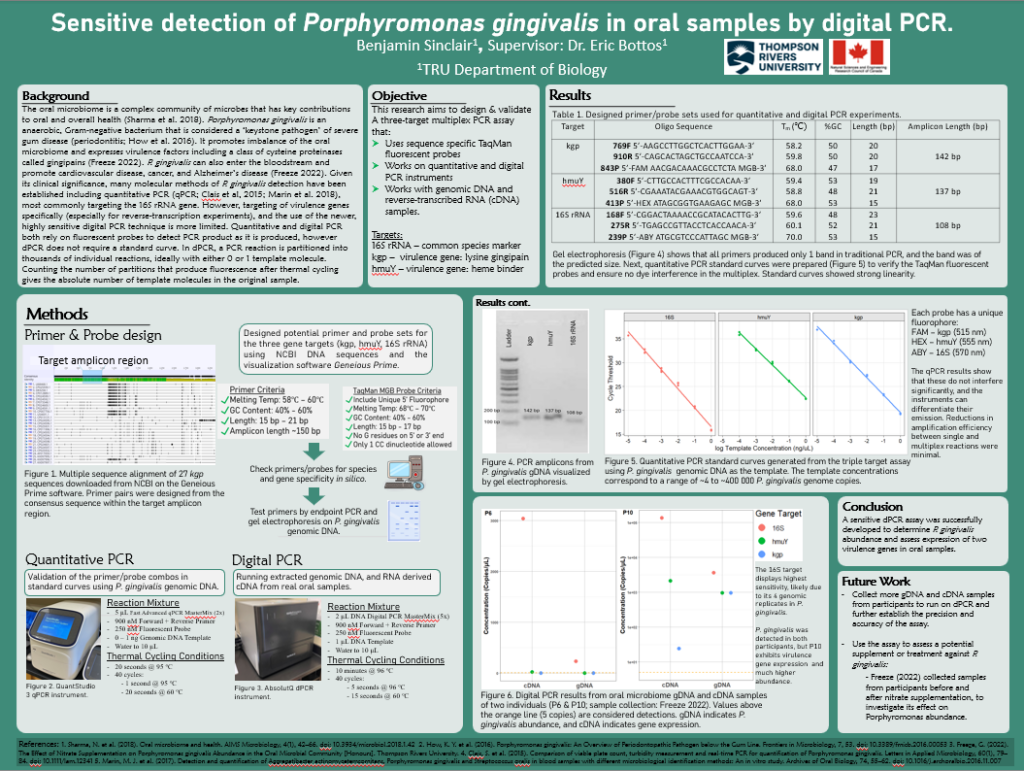
RESL 1500 Winter 2026. Thompson Rivers University.
INTRODUCTION
My name is Benjamin Sinclair and I am currently in my last semester of a BSc here at TRU, majoring in chemical biology. I have had multiple research positions during my time here and gained confidence in the research process, including formulating and testing research questions, working through challenges, and communicating results.
My first position was as a research assistant for Dr. Eric Bottos from January to August of 2024. At this time, I gained experience with basic molecular biology techniques and the process of lab research mainly by helping with other students’ projects. The next summer, I worked in the same lab under an NSERC undergraduate student research award on my own project, where I developed and validated primer sequences for three gene targets on the bacteria P. gingivalis. This project greatly increased my understanding of the research process, especially the resiliency required when inevitable barriers are met.
In the fall term of 2025, I began a directed study research project under Dr. Heidi Huttunen-Hennelly on a microbiology based experiment. This work has broadened my research experience to another topic, and presented me with new challenges of the research process. Specifically, I am continuing the work of previous students, which has many benefits but has also posed unique challenges.
Alongside these two main research projects, much of my coursework has given me practice in research skills like conducting productive background research, drawing meaningful and legitimate conclusions from data, and designing sound methodology. Group and partner lab projects in multiple courses have shown me the importance of communication and collaboration when researching, which I have brought into my other projects. All of these experiences together have given me a strong grasp on the research process including method design, data collection and analysis, and communication of findings.
THE STANDARDS
REFLECTIVE ESSAY
I’ve been at TRU for almost four years now and will complete my chemical biology degree in April of this year (2026). Recently I have had a good chance to reflect on all my experience and learning here, especially the research portion. I’ve had the opportunity to work on both molecular biology and microbiology research projects, which have significantly enriched my learning and supplemented my normal coursework. Over the course of my degree, my view of the research process has changed drastically, and I’ve developed many new skills that are necessary for conducting sound scientific research.
Going into my first year of science at TRU, I knew that I wanted to do some sort of scientific research in my future career. I was very excited that my chemistry and biology courses included lab portions and I got early experience with performing experiments. The way these experiments were presented was simplistic, where things were established to work, we knew what results to expect, the procedures were clearly outlined and given to us, and the work could be completed in a three-hour period. If the expected results were not achieved from an experiment, it simply meant the procedure wasn’t followed properly. Despite what I thought at the time, this sort of lab experience was only about getting comfortable with practical lab skills, not about conducting proper scientific research. By the end of second year, I had good experience with practical skills, and lab report writing, in different fields of chemistry and biology.
At the end of second year, I also took CMNS 2300 which taught proper conventions for writing up scientific results. I also got the opportunity to write a literature review along with a partner, which was my first big exposure to the scientific literature. I learned that evaluating and synthesizing the findings and methodologies of existing research papers is a crucial element of conducting one’s own research. Furthermore, I learned that presenting the findings of a research project to both a scientific and general audience is an important skill that all researchers should develop.
Moving into third year, I had begun to learn more about the realities of the research process through a research assistant position over the summer with Dr. Eric Bottos. Instead of being given a strict procedure and the expected results, I was given an abstract research question and not much else. The project was bioinformatics based, meaning it used only pre-existing data from online databases. I knew little about this, and it took all summer to get some insights related to the original question. I had no procedure to work from, and instead I learned how to do background research and build a methodology myself.
The structure of my third year lab classes also shifted from previous years with less defined procedures, longer experiments that lasted multiple weeks, and less certainty in expected results. Multiple classes also introduced us explicitly to a more advanced version of the research process, where we had to conduct background research to address a research question with our own methodology. These projects brought in a little bit more of the non-linear, trial and error type of work that I learned good research should include. With these projects, along with my summer work, my perspective on research shifted to be a more abstract, non-linear process where adjusting and refining a methodology is the way to get expected results. I also learned that background research, whether that be from an instructor’s lectures or the online literature, is crucial to obtain the theory needed for designing a good methodology.
After third year is when I really felt I became a student researcher. I worked in Dr. Bottos’ lab again over the summer, but this time on my own project through an NSERC USRA. This project had a clear end goal of developing a quantitative assay for detection of the bacterium P. gingivalis by PCR. At first, the steps to get there felt gargantuan, but reviewing existing literature and previous student methodology provided a good place to work from. However, there was plenty of trial and error, with the methodology going through many small tweaks and iterations before everything finally worked. One major thing I realized was the amount of creativity needed for scientific research. The next steps were often uncertain during this project, and thinking with a creative mindset was absolutely necessary. I also realized through this project that interpretation of results has much more depth than I thought previously, and an intimate knowledge of every experimental variable and their interplay is needed when drawing conclusions.
In the Fall of fourth year, I began a directed study with Dr. Heidi Huttunen-Hennelly where I hoped to use my summer experience and new perspectives to conduct some good research in a slightly different field. The first challenge I faced was writing a research proposal, including formally developing and communicating my research question. This introduced me to the “real world” part of research, where doing things like funding/award applications or for-credit directed study proposals always precedes the experimentation itself. The work on this project is microbiology based and there are many other research students doing similar work, meaning I’ve had the opportunity to talk with them to compare methodologies and results. This has been really beneficial to my project and expedited the trial-and-error process significantly, and is something I now know to be of high importance in research.
Looking back on my time at TRU, my research experience has transformed my understanding of science from structured classroom exercises into a creative, collaborative, and iterative process of experimentation. Through both my independent research and coursework, I have developed practical laboratory skills but also the ability to think critically, communicate effectively, and adapt when experiments do not go as planned. My experiences have shown me that meaningful research depends heavily on curiosity, persistence, and collaboration as well as technical knowledge. As I move forward in my career, I will carry with me the confidence and perspective I have gained as an undergraduate researcher, along with a deeper appreciation for the complexity of scientific experimentation.